 have disproportionately affected indigenous populations around the globe. Mining and milling operations in the U.S. Near tailings piles, water samples have shown levels of some contaminants at hundreds of times the government’s acceptable level for drinking water. Uranium mill tailings contain radioactive materials, notably radium-226, and heavy metals (e.g., manganese and molybdenum) which can leach into groundwater. The most serious health hazard associated with uranium mining is lung cancer due to inhaling uranium decay products. Moreover, the half-lives of the principal radioactive components of mill tailings, thorium-230 and radium-226 are long, being about 75,000 years and 1,600 years respectively. While the hazard per gram of mill tailings is low relative to most other radioactive wastes, the large volume and lack of regulations until 1980 have resulted in widespread environmental contamination. is over 95 percent of the volume of all radioactive waste from all stages of the nuclear weapons and power production. The total volume of mill tailings generated in the U.S. (In-situ leach mining leaves the unusable portion in the ground, it does not generate this form of waste). Conventional mining techniques generate a substantial quantity of mill tailings waste during the milling phase, because the usable portion is generally less than one percent of the ore. The milling (refining) process extracts uranium oxide (U 3O 8) from ore to form yellowcake, a yellow or brown powder that contains about 90 percent uranium oxide. have shut down and imports account for about three-fourths of the roughly 16 metric tons of refined uranium used domestically each year - Canada being the largest single supplier. In the past decade, alternative techniques such in-situ leach mining, in which solutions are injected into underground deposits to dissolve uranium, have become more widely used. 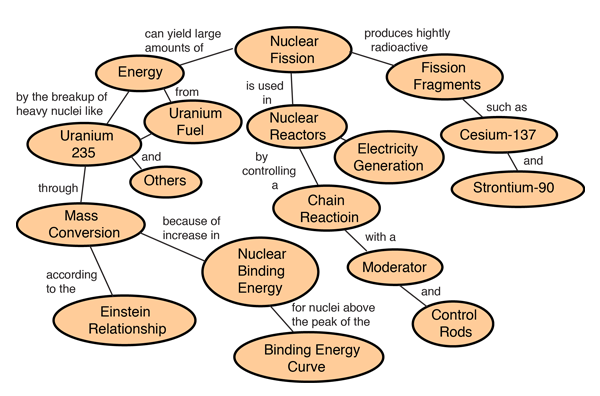
Traditionally, uranium has been extracted from open-pits and underground mines. Plutonium-239, virtually non-existent in nature, was used in the first atomic bomb tested Jand the one dropped on Nagasaki on August 9, 1945. In contrast, uranium-238 cannot sustain a chain reaction, but it can be converted to plutonium-239, which can. Of the naturally-occuring uranium isotopes, only uranium-235 can sustain a chain reaction– a reaction in which each fission produces enough neutrons to trigger another, so that the fission process is maintained without any external source of neutrons. The property of uranium important for nuclear weapons and nuclear power is its ability to fission, or split into two lighter fragments when bombarded with neutrons releasing energy in the process. Animal studies suggest that uranium may affect reproduction, the developing fetus, and increase the risk of leukemia and soft tissue cancers. Uranium is also chemically toxic at high concentrations and can cause damage to internal organs, notably the kidneys. If inhaled or ingested, however, its radioactivity poses increased risks of lung cancer and bone cancer. Uranium-238 emits alpha particles which are less penetrating than other forms of radiation, and weak gamma rays As long as it remains outside the body, uranium poses little health hazard (mainly from the gamma-rays). After several more alpha and beta decays, the series ends with the stable isotope lead-206. The various decay products, (sometimes referred to as “progeny” or “daughters”) form a series starting at uranium-238. Uranium-238 decays by alpha emission into thorium-234, which itself decays by beta emission to protactinium-234, which decays by beta emission to uranium-234, and so on.  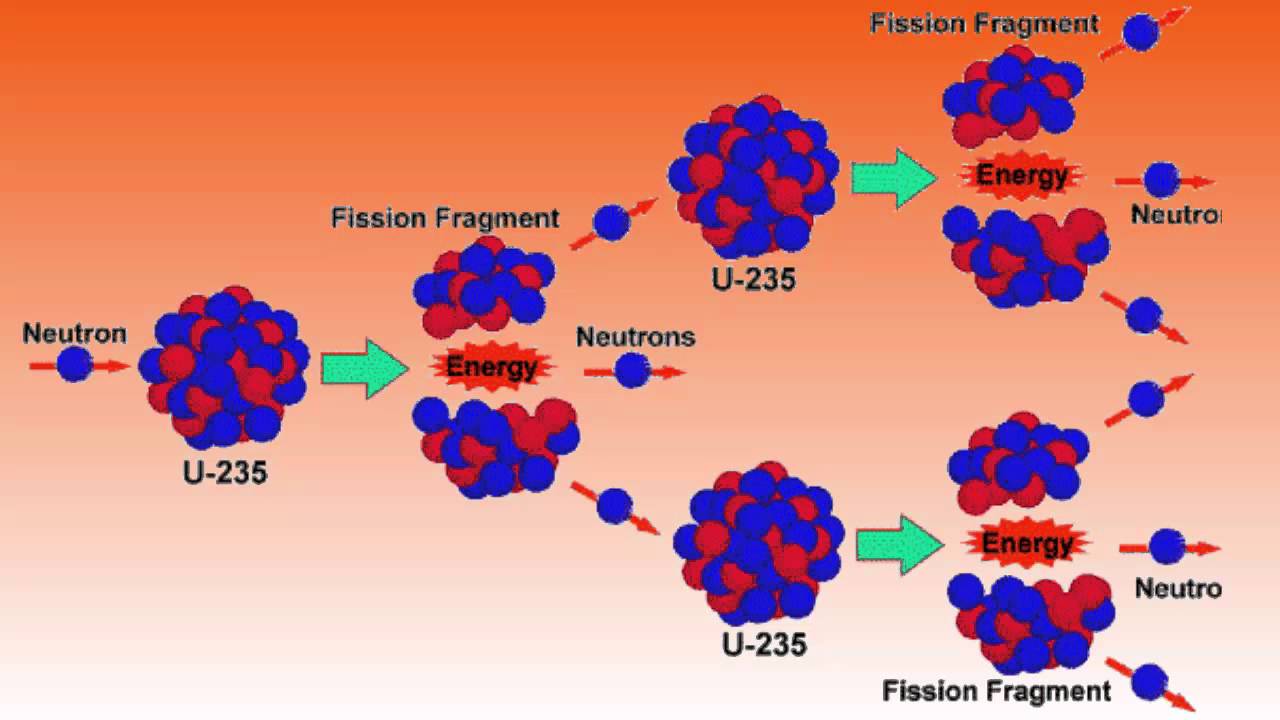
Uranium-238, the most prevalent isotope in uranium ore, has a half-life of about 4.5 billion years that is, half the atoms in any sample will decay in that amount of time. 
These three kinds of radiation have very different properties in some respects but are all ionizing radiation–each is energetic enough to break chemical bonds, thereby possessing the ability to damage or destroy living cells. It is often also accompanied by emission of gamma radiation, which is electromagnetic radiation, like X-rays. This process, known as radioactive decay, generally results in the emission of alpha or beta particles from the nucleus. The nuclei of radioactive elements are unstable, meaning they are transformed into other elements, typically by emitting particles (and sometimes by absorbing particles). Natural uranium consists of three isotopes: uranium-238, uranium-235, and uranium-234. Uranium is the principal fuel for nuclear reactors and the main raw material for nuclear weapons. In 1938, German physicists Otto Hahn and Fritz Strassmann showed that uranium could be split into parts to yield energy. Some of the terms used in this factsheet are defined in IEER’s on-line glossary.įirst discovered in the 18th century, uranium is an element found everywhere on Earth, but mainly in trace quantities. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
